|
11/25/2023 0 Comments New england journal of medicine vaping 
(Funded by the National Institute for Health Research and Cancer Research UK Current Controlled Trials number, ISRCTN60477608. There were no significant between-group differences in the incidence of wheezing or shortness of breath.Į-cigarettes were more effective for smoking cessation than nicotine-replacement therapy, when both products were accompanied by behavioral support. The e-cigarette group reported greater declines in the incidence of cough and phlegm production from baseline to 52 weeks than did the nicotine-replacement group (relative risk for cough, 0.8 95% CI, 0.6 to 0.9 relative risk for phlegm, 0.7 95% CI, 0.6 to 0.9). 51.2% in the nicotine-replacement group) and nausea more frequently in the nicotine-replacement group (37.9%, vs. Overall, throat or mouth irritation was reported more frequently in the e-cigarette group (65.3%, vs. Among participants with 1-year abstinence, those in the e-cigarette group were more likely than those in the nicotine-replacement group to use their assigned product at 52 weeks (80% vs. The 1-year abstinence rate was 18.0% in the e-cigarette group, as compared with 9.9% in the nicotine-replacement group (relative risk, 1.83 95% confidence interval, 1.30 to 2.58 P<0.001). 
The New England Journal of Medicine has published E-Cigarettes and Vaping-Related Disease, a collection of articles and other resources on e-cigarettes and vaping related diseases. Secondary outcomes included participant-reported treatment usage and respiratory symptoms.Ī total of 886 participants underwent randomization. Although vaping has been shown to assist with smoking cessation, cases of severe vaping-related pulmonary disease and a number of deaths have recently been reported. Participants who were lost to follow-up or did not provide biochemical validation were considered to not be abstinent. The primary outcome was sustained abstinence for 1 year, which was validated biochemically at the final visit. Treatment included weekly behavioral support for at least 4 weeks. National Health Service stop-smoking services to either nicotine-replacement products of their choice, including product combinations, provided for up to 3 months, or an e-cigarette starter pack (a second-generation refillable e-cigarette with one bottle of nicotine e-liquid ), with a recommendation to purchase further e-liquids of the flavor and strength of their choice. 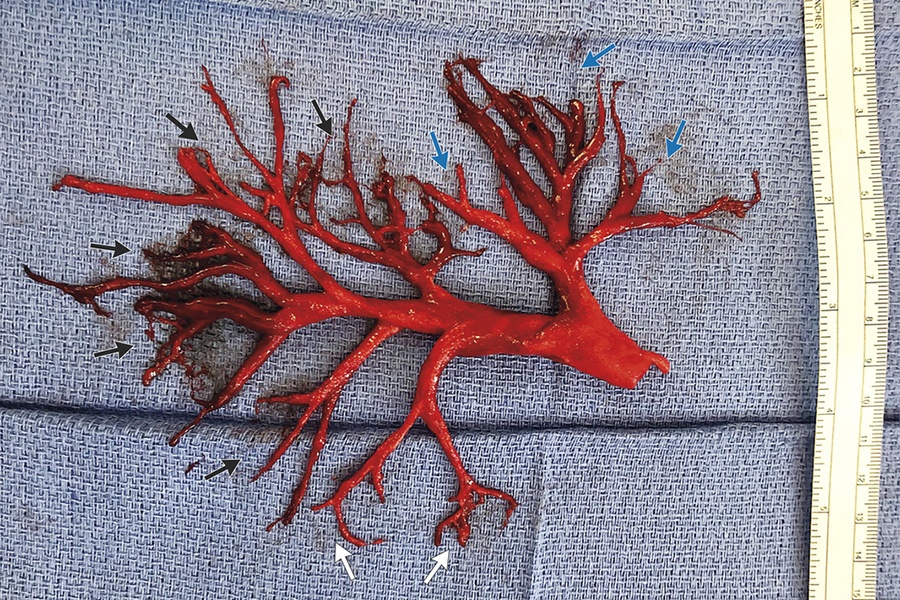
We randomly assigned adults attending U.K. E-cigarettes are commonly used in attempts to stop smoking, but evidence is limited regarding their effectiveness as compared with that of nicotine products approved as smoking-cessation treatments. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
